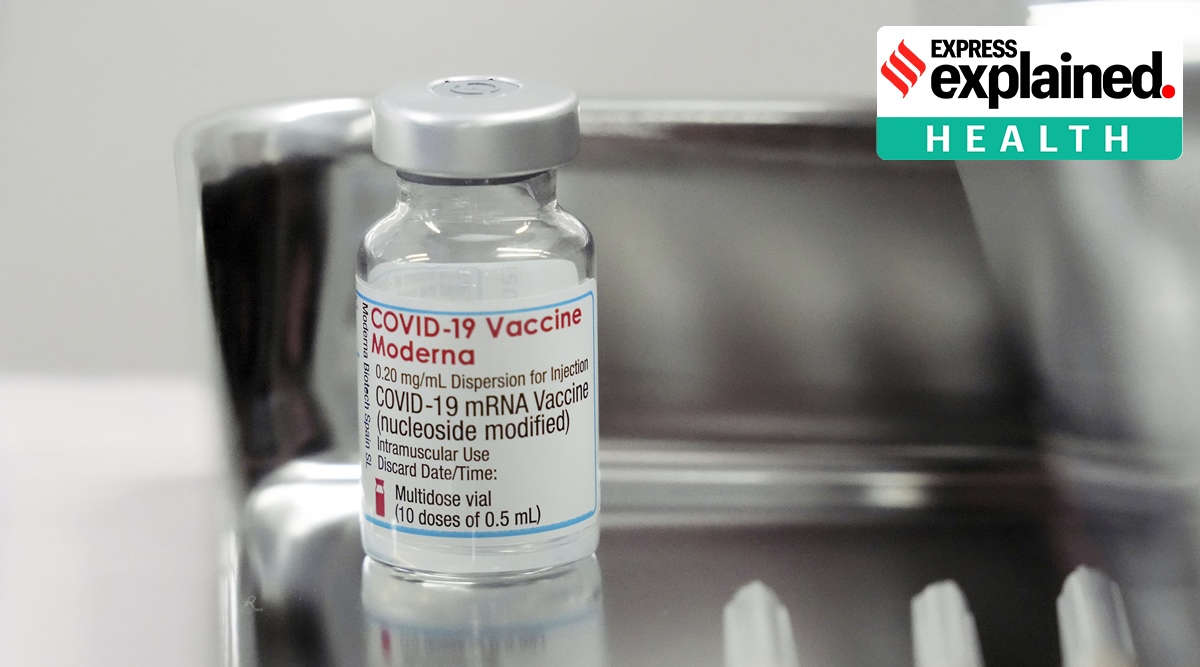
On August 26, Japan said it had halted the use of 1.63 million Moderna doses shipped to 863 vaccination centres nationwide as a precaution.
Japan’s Covid-19 vaccination push has been dealt a blow by widening reports of contamination in supplies of Moderna Inc’s (MRNA.O) Covid-19 vaccine.
Here are some key points in the issue so far.
Newsletter | Click to get the day’s best explainers in your inbox
When was it first reported and how has it widened?
The first contamination was reported to Takeda Pharmaceutical Co (4502.T), Moderna’s local distributor, on Aug. 16. That led to the discovery of 39 vials containing foreign material, all from the same lot of 57,000 vials, or about 570,000 doses.
On Aug. 26, Japan said it had halted the use of 1.63 million Moderna doses shipped to 863 vaccination centres nationwide as a precaution.

On Sunday, a vaccination centre in Okinawa prefecture reported that contaminants were found in Moderna vials from a fourth lot, 3005293.
Black substances were spotted in syringes and a vial, while pink substances were found in a different syringe, the health ministry said.
Also on Sunday, a vaccine centre in Gunma prefecture, near Tokyo, found a tiny, black substance in a Moderna vial from lot 3005236.
The affected lots from Okinawa and Gunma, accounting for about 1 million doses, were temporarily suspended.
The health ministry said on Monday it appeared that some of the cases were caused by needles being incorrectly inserted into vials, breaking off bits of the rubber stopper. Other vials from the Okinawa and Gunma lots can continue to be used, it said.
What are the contaminants?
The contaminants that triggered suspensions of the first three lots are believed to be metallic particles, public broadcaster NHK reported, citing health ministry sources.
The ministry has not officially said what the particles are, pending an investigation by Takeda, Moderna, and its Spanish contract manufacturing partner Rovi (ROVI.MC). Those companies have also yet to comment on what the foreign material is.
Is there a safety issue?
Japan and Moderna have said no safety issues had been identified.
Two men, aged 38 and 30, died this month within days of receiving their second Moderna doses, the health ministry said on Saturday. Each had one shot from one of the suspended lots, 3004734. The causes of death are being investigated.
Both men had a fever the day after their second dose and died two days after getting the fever. There has been no evidence that their shots contained contaminants, a health ministry official told reporters.
As of Aug. 8, 991 people had died in Japan after receiving Covid-19 shots from Pfizer (PFE.N)/BioNTech, and 11 after receiving Moderna’s. No causality between the injections and the deaths has been established, according to the health ministry.
About 500,000 people have had shots from the first three suspended batches, Taro Kono, the minister in charge of the vaccine push, told reporters on Friday.
Are other countries also affected?
Rovi, which bottles Moderna vaccines for markets other than the United States, said last week the batch in question was meant for distribution only in Japan.
South Korea, which also receives the Moderna vaccines produced from sites in the United States and Spain, said it was not affected.
The European Union drugs regulator said last week it was investigating the incident but did not find reasons to seek a temporary suspension of production after an initial assessment.
Source: Read Full Article